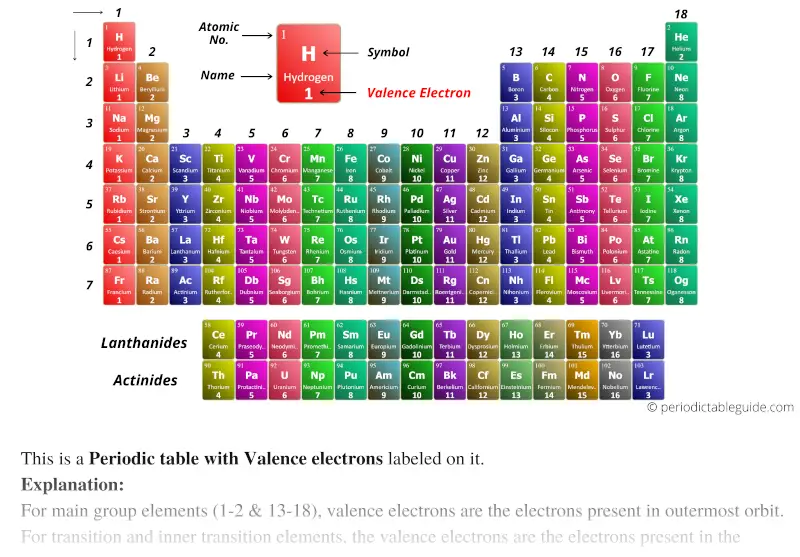
Stability is attained either by losing or gaining or sharing of electrons. Valence electrons are the electrons in the outermost shell, or energy level, of an atom. The main reason for the stability of the noble gas elements is the completely filled orbits of the atom.Įach and every element in the periodic table wants to attain the stability of noble gas as they are the most stable element in the periodic table. But the valency of elements, when combined with H or O first, increases from 1 to 4 and then it reduces to zero. While moving left to right across a period, the number of valence electrons of elements increases and varies between 1 to 8. 1: Periodic table by Dmitri Mendeleev, 1871. 
After the development of quantum mechanics, it was shown that the halogens all had seven valence electrons, supporting their original placement into the same group on Mendeleev's periodic table. Variation Of Oxidation State Along a Period. As a gas or vapor, the halogens all had a pungent odor. The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons. Periodic Trends in the Oxidation States of Elements. The periodic table and trends in valence electrons can be used to determine the number of bonds an atom is most likely to form. Atoms share electrons and form covalent bonds to satisfy the octet rule. There are total 18 vertical columns on periodic table. 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. We can see from the electron configuration of a carbon atom1 s2 2 s2 2 p2 that it has 4 valence electrons (2 s2 2 p2) and 2 core electrons (1 s2 ). As all the orbits of an atom are filled, the new electrons, if added, need a new orbit. Groups are the vertical columns on the periodic table. Elements in any one group (or column) have the same number of valence electrons (Figure 1) the alkali metals lithium and sodium each have only one valence. Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. These are at the rightmost column of the periodic table. Home Protons Neutrons Electrons Electron Configuration Orbital Diagram Valence Electrons Chemistry Calculator Your Ultimate Guide to Periodic Table Elements. Your ultimate guide to electron configuration and more Skip to content. These are named as the Noble Gas family or the inert gas. Discover the fascinating world of periodic table elements with. If every orbit in the atom is completely filled with its maximum number of electrons that it can hold then such atoms are the most stable elements. The group number defines the valence electrons present in the element. The period number represents the number of shells in an atom. 
The alkaline metals, the second group of the table, contains two valence electrons, i.e., the outermost orbit of this atom contains two electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
