The changes of state that occur with variations in temperature or pressure can be described and predicted using the kinetic molecular theory.We also assume that we have determined a complete set of. The elements consist of identical atoms, and compounds consist of identical molecules, which are particles containing small whole number ratios of atoms. The relative amount of kinetic energy in a group of atoms or molecules is an important factor in determining its physical state. We assume an understanding of the atomic molecular theory postulates, including that all matter is composed of discrete particles.The kinetic molecular theory states that atoms and molecules are perpetually in motion and have kinetic energy.Elements in the same row of the periodic table contain the same number of energy levels.These electrons are called valence electrons and give rise to similar chemical properties.Elements in the same vertical column or group of the periodic table contain the same number of electrons in their outer energy levels.The organization of the periodic table can be used to predict the metallic character and tendency of main group elements to form ionic or covalent bonds.The atom consists of subatomic particles that differ in location, charge, and relative mass.The properties of an atom are based on the number and arrangement of its parts.Atoms are the basic building blocks of all matter.Different substances with different properties are suited to different uses.Matter consists of atoms held together by electromagnetic forces matter exists as different substances which can be utilized based on their properties. An atom is the smallest unit of matter that still has all of the properties of its element, whereas a molecule is a structure that contains multiple atoms.Note: the Bohr model is an inaccurate model and does not depict the 3-D nature of the atom it implies that electrons are in static orbits.The electron cloud model describes the atom as containing a dense nucleus of protons and neutrons surrounded by regions of space (clouds) where electrons are most likely to be found.The electron cloud model best represents our current understanding of the atomic structure.The development of this theory illustrates the nature of science.The atomic theory encapsulates our current understanding of the atom and its structure. 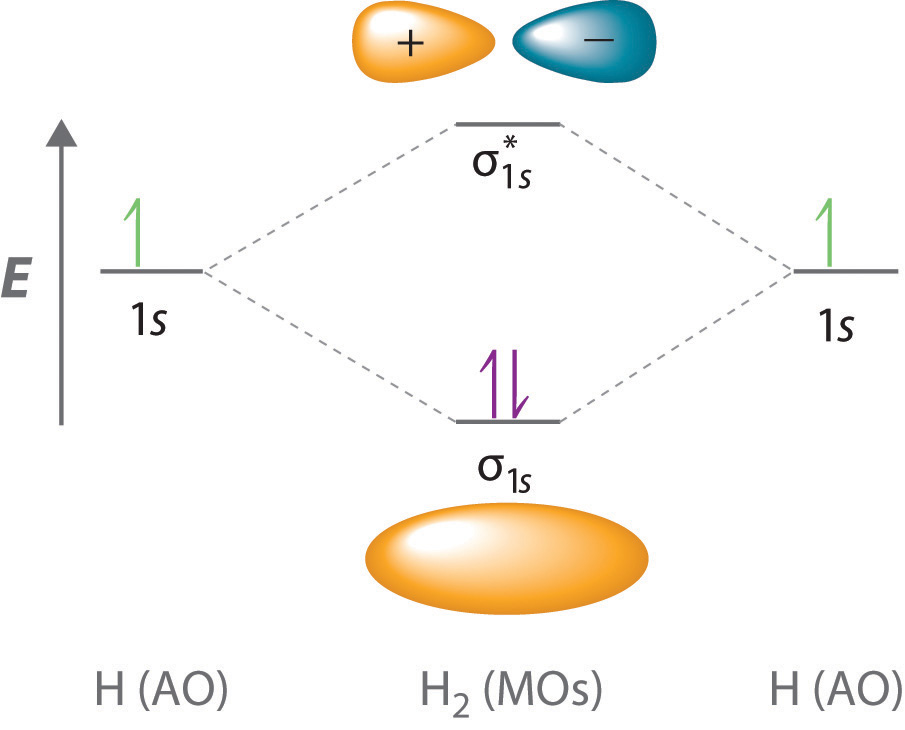
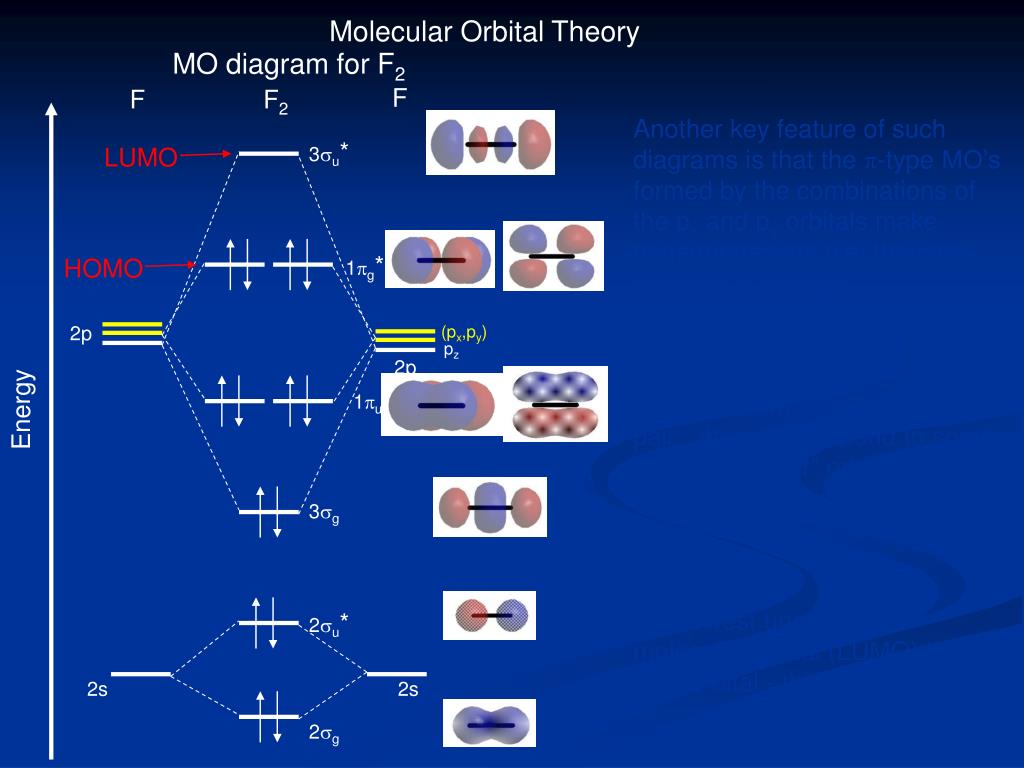
This module shows how the mole, known as Avogadro’s number, is key to calculating quantities of atoms and molecules. will exist, explain many properties for example why O2 is a paramagnetic diradical and identify the important frontier molecular orbitals (FMOs). Even rather simple molecular orbital (MO) theory can be used to predict which homonuclear diatomic species H2, N2, O2, etc. A series of contributions and discoveries has led to the development of the atomic theory. In science, atomic theory is an argument that all matter in the universe consists of small particles called atoms or atomos (Greek meaning uncuttable or. The mole is an important concept for talking about a very large number of things 6.02 x 10 23 of them to be exact. Diatomic Species by Molecular Orbital Theory.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
